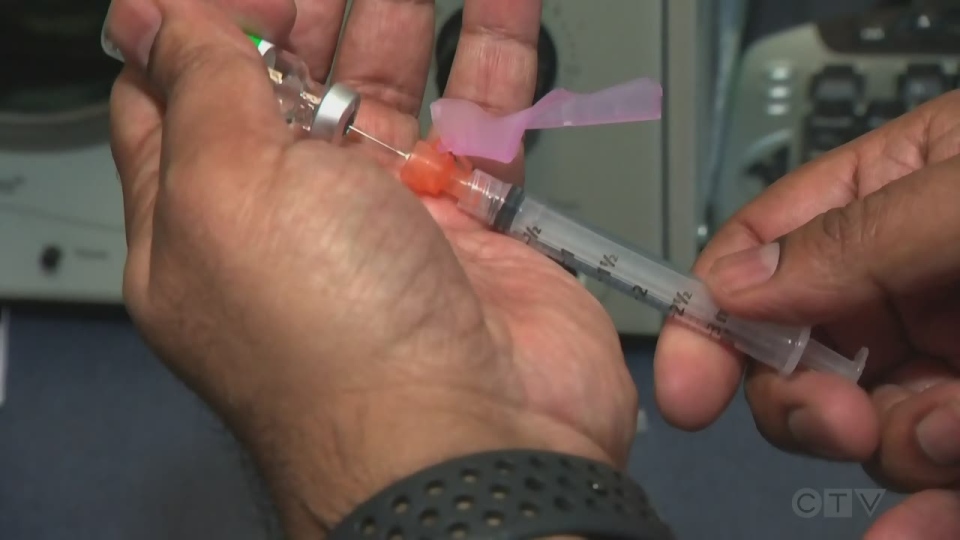
GUELPH -- Parents are anxiously awaiting more information about a possible COVID-19 vaccine approval for children under the age of 12.
“We're happy that he's going to school but we're a little nervous that he's unprotected,” Rob Penfold told CTV News while walking his six-year-old son home from school in Guelph Wednesday. “So the sooner the vaccine is available for his age group, the better.”
Many parents said they like the idea of vaccinating their young children but won’t make a final decision until they hear more about the medical research.
“How will it affect the kids in a longer time? As far as their health is concerned, as far as their growth is concerned,” wondered Monika Parmar, also the parent of a school-aged child.
On Monday, Pfizer released preliminary information from a phase 2/3 trial of their COVID-19 vaccine for children between the ages of 5 and 11. According to a company press release the trial involved “a two-dose regimen of 10 µg administered 21 days apart, a smaller dose than the 30 µg dose used for people 12 and older.”
“As [trial participants] are getting down littler and littler, we’re seeing littler and littler doses but what they’re looking to see is to make sure it’s the same amount of antibodies regardless of the dose you got,“ said Kelly Grindrod, a member of the Region of Waterloo Vaccine Taskforce and a professor at the University of Waterloo’s School of Pharmacy. “That’s what these trials are actually looking at.”
Summarizing the trial results, the Pfizer release stated, "in participants 5 to 11 years of age, the vaccine was safe, well-tolerated and showed robust neutralizing antibody responses."
“They say that it’s safe which is great news,” Grindrod added. “This is actually very good news because it tells us that they're at the stage where we can start thinking about the approval of the vaccines for children but we know very little about what the data actually says so far.”
South of the border, Pfizer is seeking authorization for the emergency use of its COVID-19 vaccine in American children ages 5 to 11.
Pfizer Canada told CTV News Kitchener they share an "urgency" to provide that data to Health Canada but were unable to provide a timeline of when they will formally submit it for approval.
As for the logistics of how shots will get into arms, the rollout may be organized by each individual Health Unit.
“We are working closely with local physicians to administer vaccines to this age group as soon as they’re approved for them," Wellington-Dufferin-Guelph Public Health said in a statement.
WDG Public Health also stated the plan for this age group would likely see a combination of vaccinations done through appointments at mini-mass clinics, in doctor’s offices and in public health offices but reiterated the fact that plans are not yet concrete.
Region of Waterloo Public Health said it is likely to continue the use of existing vaccine clinics but with a family twist. They are looking to make changes, taking lessons learned from clinic survey data.
The health unit is also looking into the possibility of organizing after-school clinics.
"Key partners, such as primary care and community pharmacies are collaborating with us on a variety of options up for consideration," said Sharon Ord, vaccine communications lead for Region of Waterloo Public Health.
CTV News reached out to local school boards asking if they are working with health units to possibly administer shots but were directed back to public health officials.
Ontario’s Ministry of Health said the province “continues to follow NACI’s guidelines that individuals who are born in 2009 or earlier are eligible to receive the COVID-19 vaccine.”
In a written statement, the Ministry added they are continuously monitoring evidence and are following the lead of Health Canada.
“Working with our public health and health system partners we will be ready to administer doses to children aged five to 11 as soon as they are approved by Health Canada," the Ministry said.
But when could a vaccine be approved?
“It all depends what the data shows. They go over this data especially for kids extremely carefully,” Grindrod explained.
There have been speculations the United States may begin vaccinating younger children by the end of October, but Grindrod hinted that timeline seems a little ambitious for Canadians given Pfizer hasn’t officially submitted its data for approval yet.
“I think we have some confidence now it’s going to be likely before Christmas, likely before the new year. The question is when. Is it October? November? We don’t really know yet,” Grindrod said.
Pfizer says trial results for children under the age of five may be available as soon as the end of the year.